|
In this page we will discuss the wave properties of electrons and how it has advanced our understanding of the atom.
Quantum Mechanical Model of the AtomOur most current understanding of the atom is the most confusing yet. Fear not, students of chemistry, some of our generations most brilliant minds are studying this theory to better understand it!
One of the most confusing pillars of quantum theory is understanding how electrons can behave as both a wave and a particle. Dr. Richard Feynman once said about quantum mechanics: |
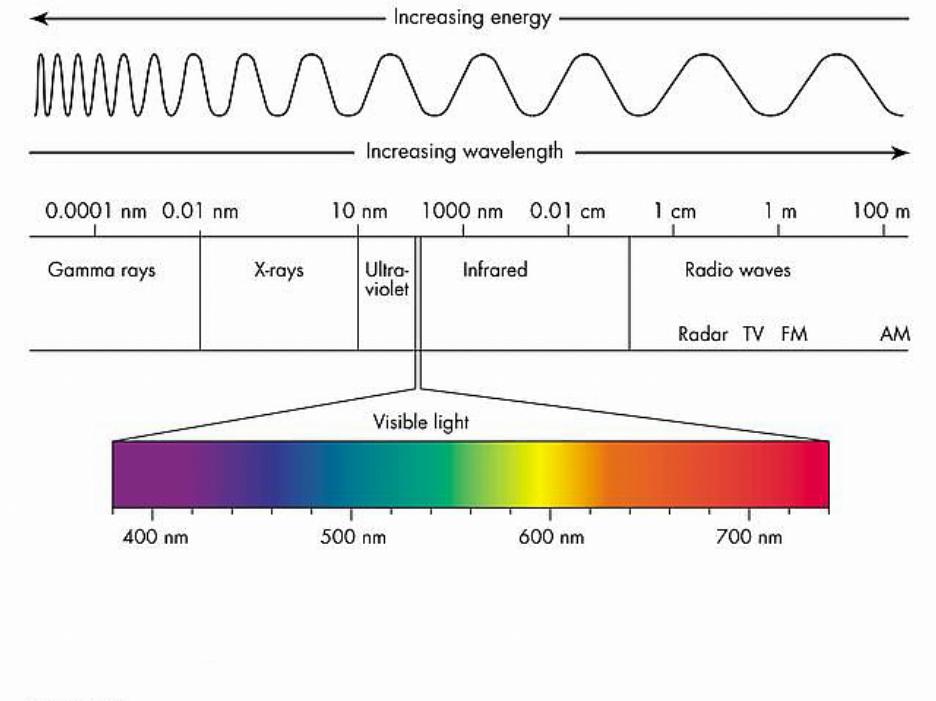
Nature of LightLight has a split personality: sometimes it behaves like a wave and other times it behaves like a particle. The colors of light that we see are a result of the wave-like property of light (see the picture below). Understanding light and how it behaves is fundamental to our understanding of the quantum mechanical model of the atom.
Pilot-Wave TheoryLouis deBroglie pioneered the theory of electrons behaving both as waves and particles (like photons of light) called a pilot-wave. Veritasium has put together a really cool video that attempts to demonstrate deBroglie's pilot-wave theory that you can watch below.
|