|
In this page we will look at how matter is defined and classified.
Physical Vs. Chemical ChangesEvidence that a change has occurred:There are several observations that can be made to suggest that a chemical (rather than physical) change has occurred. These are not absolute, and you may still observe these occurrences during a physical change.
|
Mass, Volume, and DensityMass is a measure of the amount of matter in an object.
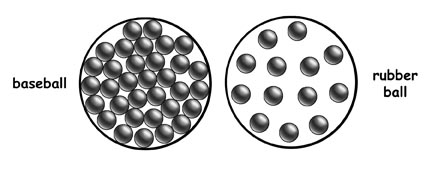
Volume is a measure of the amount of space an object takes up. Density is the ratio of the mass to the volume; it can be thought of as how closely packet the matter is that makes up the object. This image above represents the differing densities of a baseball and a rubber ball. If both objects take up the same amount of space (volume) and the baseball contains more matter (mass), then we can determine that the baseball's particles are more closely packed (density) compared to the rubber ball.
Often times mass and weight are confused, click the button below to see a video from NASA explaining the difference.
Intensive vs. Extensive Properties of MatterSimply put,
|